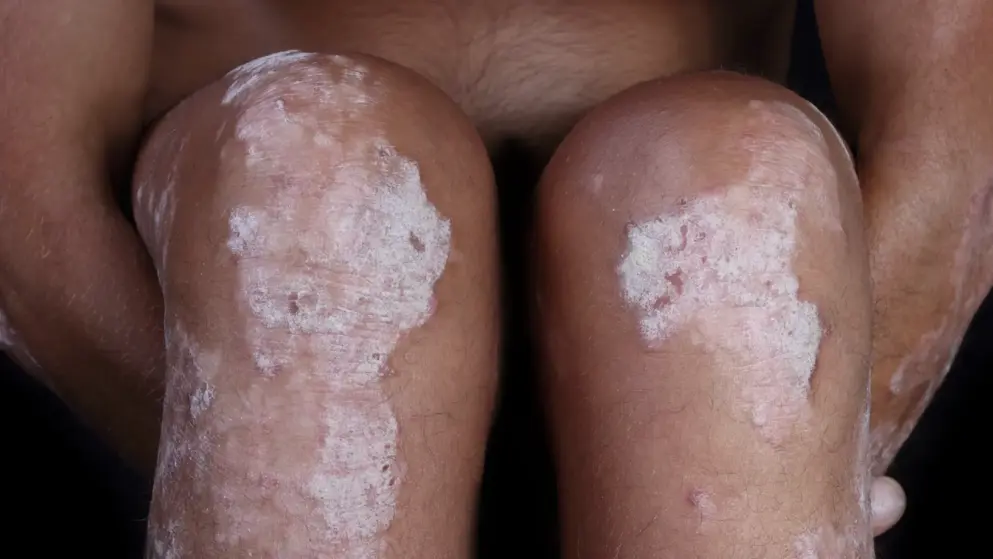
FDA action for BLA for bimekizumab for the treatment of adults with moderate to severe plaque psoriasis is now shifted to Q3 2023.-UCB
UCB, a global biopharmaceutical company,announced that the Biologics License Application (BLA) for bimekizumab for the treatment of adults with moderate to severe plaque psoriasis remains under review with the FDA.
UCB previously communicated the FDA action was expected in Q2, 2023. UCB now anticipates the FDA action in Q3, 2023. There are no open Information Requests from the FDA regarding the BLA for bimekizumab.
Bimekizumab, an IL-17A and IL-17F inhibitor, is currently approved for moderate to severe psoriasis by 10 regulatory authorities and in 39 countries worldwide. In June 2023, in countries of the European Union/European Economic Area, bimekizumab was approved for two additional indications – the treatment of adults with active psoriatic arthritis, and for the treatment of adults with active axial spondyloarthritis (axSpA), including non-radiographic axSpA and ankylosing spondylitis, also known as radiographic axSpA.